

Radiocarbon Dating
Detailed
information
Detailed information about the method of radiocarbon dating, the
active laboratories and the literature is
available on the Radiocarbon website.
Equilibrium ratio
Radiocarbon dating is based on the presence of the an equilibrium ratio of
R(0) = 14C/12C in the carbon cycle. Organisms take up
carbon during their life and so obtain the equilibrium value from the
environment. During the growth the carbon and its ratio becomes fixed
in cells of plants, animals and humans. After the
death of the organism the exchange with the environment stops and also the
uptake of 14C. Because of the decay of
14C, the
14C/12C
ratio decreases according to R(t)=R(0) exp(-t/t1/2), where t1/2
is the half life. So, the ratio R(t) is a measure for the age of the material.
Reservoir age
The equilibrium ratio is not everywhere the same on earth. This causes apparent
age differences, known as reservoir ages. In a marine environment, the
average reservoir age is 400 years, because of the dissolution of old
carbonates from sediment. Such reservoir ages can also be present in lakes,
rivers and other carbonates. Other complications exist, which are connected
with the possibility to select the correct chemical fraction without
contamination (see material).
Radiocarbon age
The time past since death is derived from the measured ratio R(t)
with t = - 8033 ln [R(t)/R(0)].
The uncertainty Dt of the age t depends on the uncertainty of
R(t) through Dt = 8033 DR/R, which shows that an 1% uncertainty in R results in an 80
year uncertainty of age. Although the carbon dioxide mixes well in the
atmosphere variations in the
equilibrium value R(0) occur. are not taken into account. To correct for these a calibration
curve is used which is based on the 14C-ages of tree-rings of
known calendar age as identified from dendrochronology.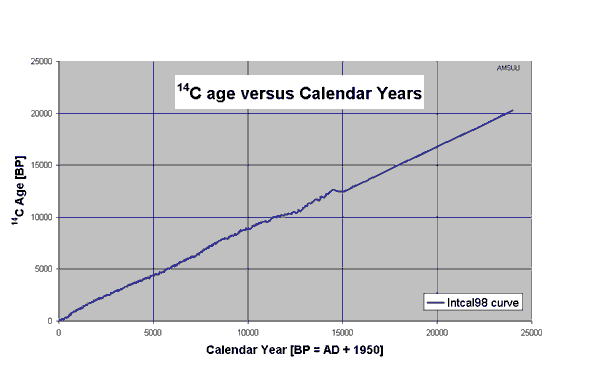
Calibration to calendar years
Calibration of the interval spanned by the 14C-age and its
uncertainty to a corresponding interval can result in one or more valid
intervals. This depends on the smoothness of a calibration curve. For
example, 14C dates from the period after 1660 AD cannot be
discriminated against modern material, as is shown in the figure. Various
calibration programs are available to calculate the calendar age interval
from the 14C date.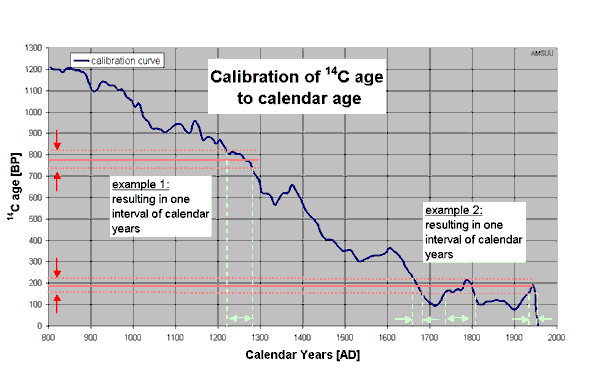
The
14C bomb peak
Nuclear weapon testing in the atmosphere caused in the 1960's almost a
doubling of the 14C activity in the atmosphere. After the
stop of these nuclear tests in the atmosphere the peak started to decrease
through interaction of the atmosphere with the other carbon reservoirs.
The
14C data showing the bomb peak on the northern and southern
hemisphere reflect the exchange between both hemispheres nicely. The rise
of the peak on the northern hemisphere, where the tests were made, is only
a year earlier than on the southern hemisphere. Clearly, the
14C-bomb
peak in both hemispheres can be used to retrieve very precise
dates in this period.