

Method
The principle of AMS is the
identification of a few individual atoms of an isotope amidst an enormous
abundance of its stable isotope. This occurs with an AMS setup. The
prepared material is loaded in the ion source, which converts material
into ions.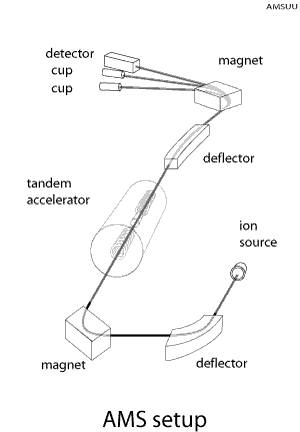
Ions are selected on mass with a bending magnet and injected into a particle accelerator, where they become accelerated. Ions emerging the accelerator are selected on energy and mass and finally detected. The stable isotope is collected in a Faraday cup and its yield is determined from the accumulated charge. The rare isotope is identified and counted in a detector.
The isotopes are measured sequentially. A fast switching between the isotopes is used for reducing transmission variations between isotopes. The large sensitivity of the AMS method originates from a combination of specific ionization properties of atoms and molecules and nuclear physics detection techniques.
The sensitivity allows measurement of isotope ratios down to 10-15. Especially cosmogenic radio nuclides, which are formed under influence of cosmic rays, and occur in very small concentrations, can be used in a broad range of applications. In radiocarbon dating AMS has extended the classical decay-counting technique through the use of small samples, milligrams in stead of grams.